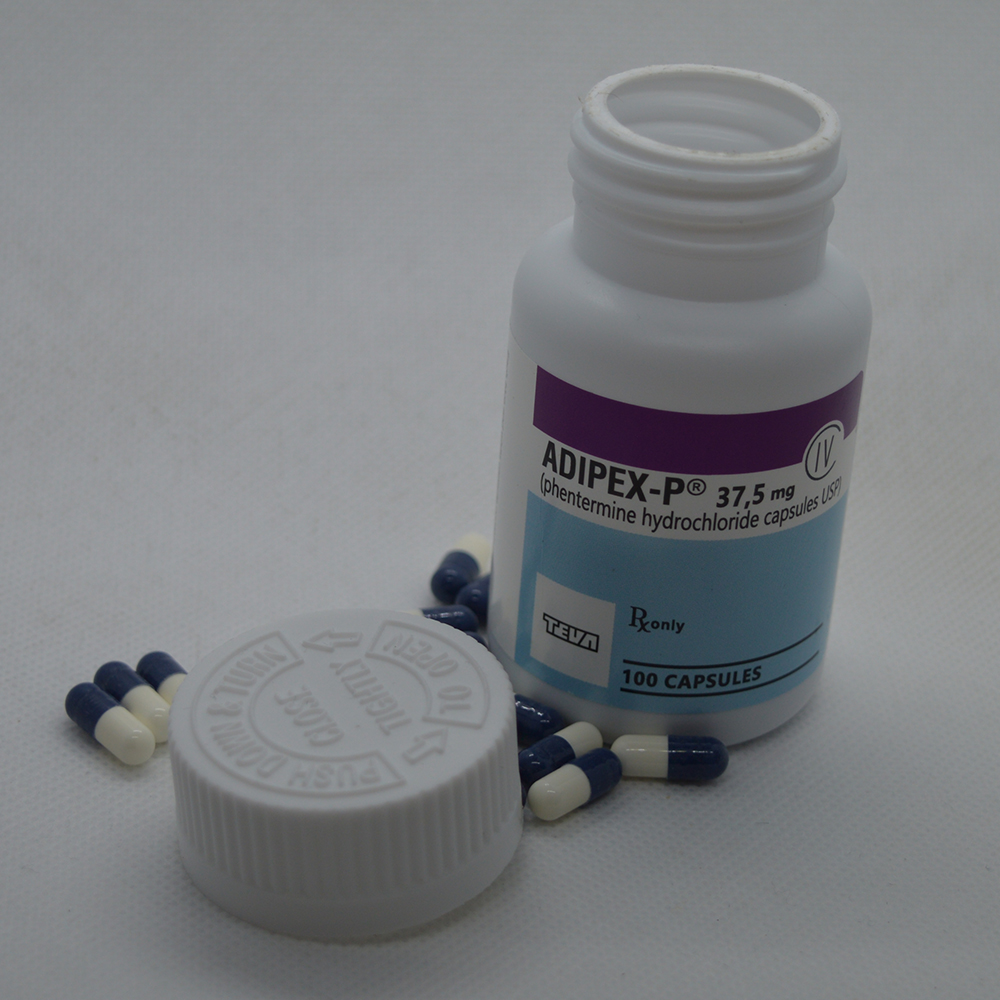
Product Description
Adipex-P represents a well-established pharmaceutical agent in the field of weight management, primarily recognized for its active ingredient, Phentermine. As a prescription-strength anorectic, it is designed for individuals requiring clinical intervention to achieve significant weight reduction. At Health Aid Pharmacy, we provide access to this compound strictly for medical reference and research applications, ensuring that professionals and researchers have a reliable source for their investigative work.
So, what exactly is Adipex-P? It belongs to a class of drugs known as sympathomimetic amines, functioning as a central nervous system stimulant. Its primary mechanism is the suppression of appetite by influencing the hypothalamic region of the brain. Specifically, it triggers the release of neurotransmitters such as norepinephrine, which signals the body’s “fight or flight” response, effectively reducing hunger cues. For research purposes, this mechanism is critical for studying metabolic pathways, obesity interventions, and energy expenditure dynamics.
The advantages of utilizing Adipex-P in a controlled research setting are notable. Its efficacy as a short-term adjunct in weight loss protocols is well-documented, making it a benchmark compound in comparative studies against newer weight-loss agents. Researchers value its rapid onset of action and predictable pharmacokinetic profile. The compound is typically indicated for patients with a body mass index (BMI) of 30 or higher, or those with a BMI of 27 or higher who present with weight-related comorbidities such as hypertension or type 2 diabetes. In a laboratory environment, this allows for the study of drug interactions and the physiological impacts of sustained appetite suppression.
Responsible usage and safety considerations are paramount when handling Adipex-P. As a controlled substance, it is intended for short-term use—typically up to 12 weeks—to avoid the potential for tolerance and dependency. Safety protocols require strict monitoring for adverse effects, which can include elevated blood pressure, increased heart rate, restlessness, or insomnia. It is contraindicated in individuals with a history of cardiovascular disease, hyperthyroidism, glaucoma, or a history of substance abuse. For research integrity, it is essential to adhere to these safety parameters to ensure the validity of study outcomes and the welfare of test subjects.
Regarding longevity and storage life, Adipex-P demonstrates excellent stability when stored correctly. The shelf life is typically 24 to 36 months from the date of manufacture, provided the product is kept in its original, airtight packaging. To preserve its chemical integrity, it must be stored in a controlled environment at room temperature (between 68°F and 77°F or 20°C and 25°C), away from moisture, heat, and direct light. Exposure to extreme temperatures or humidity can degrade the active ingredient, compromising its efficacy for research.
The composition of Adipex-P centers on Phentermine Hydrochloride. The formulation is often presented as a resin complex or immediate-release capsule, allowing for either sustained or rapid absorption depending on the specific research requirements. The standard concentration is 37.5 mg, which represents the highest commonly available dosage for clinical use, serving as a potent tool for dose-response studies.
In the context of research, Adipex-P is utilized to explore a wide array of scientific questions. These usage areas include behavioral pharmacology related to feeding patterns, cardiovascular responses to anorectic agents, and comparative efficacy studies against novel GLP-1 agonists. For institutions and laboratories dedicated to advancing the understanding of metabolic disorders, having a reliable supply of this reference standard is crucial. We invite you to explore our catalog to support your next research initiative. For more information on our full range of pharmaceutical research tools, please visit our homepage.







There are no reviews yet.