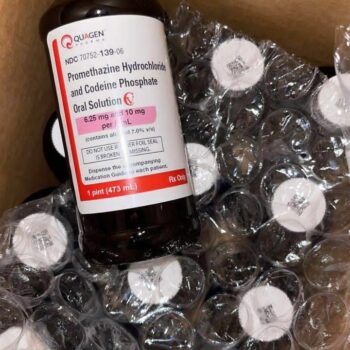
Understanding Actavis Promethazine Codeine Cough Syrup: A Professional Overview for Research and Medical Reference
In the landscape of pharmaceutical history and respiratory therapeutics, few formulations have garnered as much attention in both medical literature and public discourse as Actavis Promethazine Codeine Cough Syrup. At Health Aid Pharmacy, we believe in fostering a deep, educational understanding of pharmaceutical products to support research, medical history, and professional insight. It is crucial to approach this topic with the respect and caution it demands, focusing on its pharmacological profile rather than any extra-medical use.
What Is It and How Does It Work?
Actavis Promethazine Codeine Cough Syrup is a prescription-only medication that combines two primary active ingredients to create a powerful therapeutic effect for specific respiratory conditions. The first is Codeine Phosphate, an opioid alkaloid that acts directly on the cough center in the brain (the medulla). By depressing the cough reflex, it effectively reduces the frequency and intensity of non-productive (dry) coughing. The second is Promethazine Hydrochloride, a phenothiazine-derivative antihistamine. Promethazine serves multiple functions in this formulation: it dries upper respiratory secretions (like runny nose and watery eyes), acts as a mild sedative to allow for rest, and enhances the cough-suppressing action of the codeine .
Historically, this combination was indicated for the temporary relief of coughs and upper respiratory symptoms associated with allergies or the common cold in appropriate patient populations . The synergy between the two compounds provided a multi-faceted approach to symptom management that single-ingredient products often could not achieve.
Benefits and Historical Advantages
For the patient populations for whom it was deemed appropriate by a physician, the benefits of this formulation were notable. The primary advantage was its dual-action formula. While the codeine targeted the neurological trigger for coughing, the promethazine worked locally and systemically to manage the histamine response, reducing the post-nasal drip and congestion that often exacerbate coughing fits. Additionally, the sedative properties of promethazine helped patients rest, which is often critical for recovery from illness.
For research purposes, studying this formulation offers insights into:
-
Opioid-based antitussive therapy.
-
Combination therapies for symptomatic relief.
-
The pharmacokinetics of central nervous system (CNS) depressants.
Composition and Ingredients
The standard formulation of the original Actavis product, as documented in FDA records, contained specific concentrations designed for measured dosing. Typically, each 5 mL (one teaspoonful) contained Codeine Phosphate (10 mg) and Promethazine Hydrochloride (6.25 mg) . The base also included inactive ingredients such as alcohol (historically around 7%), along with flavorings, preservatives (methylparaben, propylparaben), and sweeteners to aid palatability .
Safety and Responsible Usage Considerations
This is the most critical section of any discussion regarding this product. Actavis Promethazine Codeine is a Schedule V (C-V) controlled substance due to its codeine content, which carries risks of dependence, misuse, and addiction.
-
Contraindications: It is vital to note that this medication is contraindicated in pediatric patients (specifically those under 6 years of age, and warnings exist for those under 18), individuals with breathing problems (like asthma or COPD), and those with known hypersensitivity to any of its ingredients .
-
Respiratory Depression: The most serious risk is respiratory depression. Codeine can suppress the drive to breathe, and promethazine can compound this effect. This risk is particularly pronounced in children and in individuals who are “ultra-rapid metabolizers” of codeine (a genetic trait that causes the body to convert codeine into morphine faster and more completely than normal) .
-
Regulatory Status: Due to widespread recreational abuse (known as “lean,” “purple drank,” or “sizzurp”), Actavis made the unprecedented decision to discontinue manufacturing its brand of this syrup . While generic versions may exist, the original Actavis brand is no longer in commercial production, making it a subject of historical and pharmaceutical research rather than an actively prescribed item in many regions.
Longevity and Storage Life
For research specimens or historical reference, the shelf life of this syrup, when stored properly, was generally 24 to 36 months from the date of manufacture, as indicated on the packaging. To maintain chemical stability and prevent degradation of the active ingredients, storage conditions are paramount. The product must be kept in a tightly closed, light-resistant container at a controlled room temperature (typically 68°F to 77°F / 20°C to 25°C). It should be protected from freezing, excessive heat, and moisture .
Usage Areas for Research Purposes
Today, the primary relevance of Actavis Promethazine Codeine lies in academic and professional research. This includes:
-
Historical Pharmacology: Studying the evolution of cough suppressants and the shift away from opioid-based therapies.
-
Public Health Studies: Analyzing the epidemiology of prescription drug abuse and the societal impact of glamorized drug use in media .
-
Regulatory Science: Understanding how the FDA and pharmaceutical companies respond to the misuse of medications.
-
Clinical Toxicology: Researching the effects and detection methods for opioid/antihistamine combinations.
At Health Aid Pharmacy, we provide this information to support the global community of researchers, medical writers, and healthcare historians. We emphasize that this content is for educational purposes only and is not a solicitation for the purchase of this controlled medication. The trust of our readers is built on the foundation of accurate, responsible, and safe communication regarding all pharmaceutical products.








There are no reviews yet.