Quagen Promethazine with Codeine Cough Syrup: A Comprehensive Overview for Medical Reference
In the landscape of respiratory therapeutics, few formulations have demonstrated the enduring efficacy of the antihistamine-opioid combination. Quagen promethazine with codeine cough syrup represents a pharmaceutical standard for the management of specific upper respiratory conditions. At Health Aid Pharmacy Inc., we provide access to this prescription-grade solution for medical and research applications, ensuring that professionals understand both its therapeutic profile and its pharmacological responsibilities.
What the Product Is
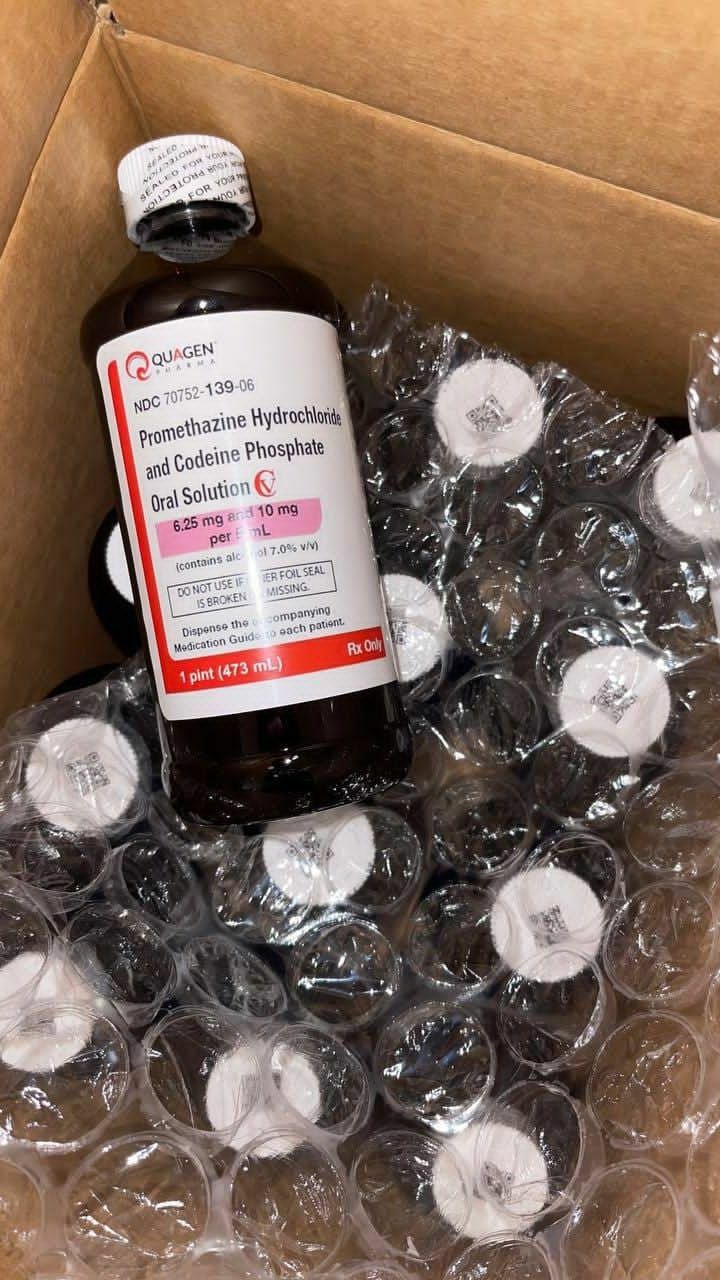
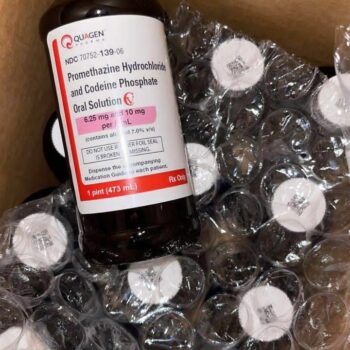
Quagen (promethazine hydrochloride and codeine phosphate oral solution) is a prescription-only, controlled substance (Schedule CV) combination medication. It is supplied as a purple-colored, raspberry-flavored oral solution, meticulously manufactured by Quagen Pharmaceuticals LLC . Each 5 mL of this solution delivers a precise dose of 6.25 mg of promethazine hydrochloride and 10 mg of codeine phosphate, suspended in a flavored syrup base . It is specifically indicated for the temporary relief of coughs and upper respiratory symptoms associated with allergies or the common cold in patients 18 years of age and older .
How It Works
The formulation’s efficacy is derived from its dual mechanism of action, targeting symptoms from two distinct neurological pathways. Codeine phosphate, an opioid agonist, works centrally by binding to opioid receptors in the cough center of the medulla oblongata. This action elevates the cough threshold, effectively suppressing the urge to cough . Concurrently, promethazine hydrochloride, a phenothiazine-derivative antihistamine, provides symptomatic relief by blocking histamine (H1) receptors. This action reduces the systemic effects of allergic reactions, including runny nose, sneezing, and itchy, watery eyes, while also contributing to the overall sedative effect that can be beneficial for patients requiring rest .
Benefits and Advantages
For the appropriate adult patient population, this combination offers significant therapeutic advantages. By integrating an antitussive with an antihistamine, Quagen promethazine with codeine provides a multi-symptom approach in a single formulation. This synergy not only suppresses non-productive, irritating coughs but also alleviates the accompanying upper respiratory congestion and discomfort typical of severe colds or allergies. The raspberry flavoring enhances patient compliance, making administration easier for those who may be sensitive to bitter medicinal tastes .
Safety and Responsible Usage Considerations
It is imperative to approach this formulation with the highest regard for safety, given its opioid component. Quagen promethazine with codeine is contraindicated in children younger than 12 years, and in children younger than 18 years following tonsillectomy or adenoidectomy due to the risk of life-threatening respiratory depression . For adults, usage must be precise: 5 mL every 4 to 6 hours as needed, not exceeding 30 mL in 24 hours . Patients must use an accurate milliliter measuring device—never a household teaspoon—to avoid accidental overdose . This medication can cause marked drowsiness, dizziness, and impaired judgment; patients should avoid driving, operating machinery, or consuming alcohol and other CNS depressants (like benzodiazepines) while under treatment .
Longevity and Storage Life
To maintain its chemical stability and therapeutic efficacy, proper storage is essential. The solution should be stored at controlled room temperature, typically between 20°C to 25°C (68°F to 77°F), protected from light, moisture, and excessive heat. When stored as directed by a licensed pharmacist in its original amber bottle—designed to block UV light degradation—the solution maintains its potency through the expiration date indicated on the package . Always check the “use by” date prior to administration.
Ingredients / Composition
The precise composition of Quagen promethazine with codeine ensures consistent pharmacokinetics. The active ingredients are Codeine Phosphate (10 mg/5 mL) and Promethazine Hydrochloride (6.25 mg/5 mL). The inactive ingredients form a stable, palatable vehicle, including: anhydrous citric acid, ascorbic acid, D&C Red No. 33, edetate disodium, alcohol, FD&C Blue No. 1, glycerin, methylparaben, propylparaben, purified water, saccharin sodium, sodium benzoate, sucrose, and trisodium citrate dihydrate .
Usage Areas for Research Purposes
For researchers and medical professionals, this product serves as a critical reference standard for studying opioid antitussives and first-generation antihistamines. It is utilized in analytical chemistry for method development, in formulation science to study syrup stability and bioavailability, and in clinical research evaluating respiratory depression and drug interaction profiles (such as CYP2D6 metabolism variability) . It remains a controlled substance, and all handling must comply with DEA regulations.
By choosing Health Aid Pharmacy Inc., you are partnering with a distributor that prioritizes pharmaceutical integrity, patient education, and global compliance.







There are no reviews yet.