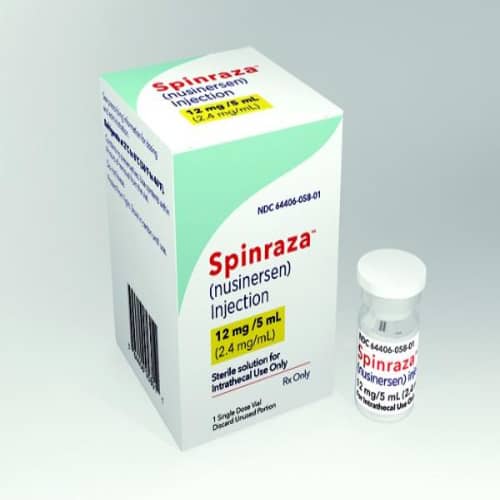
Spinraza (Nusinersen) – Advanced Treatment for Spinal Muscular Atrophy
Spinraza (nusinersen) is a groundbreaking, prescription-only therapy designed to treat Spinal Muscular Atrophy (SMA), a rare and often life-threatening genetic disorder that affects motor neurons and leads to progressive muscle weakness. As one of the first disease-modifying treatments approved for SMA, Spinraza has transformed patient outcomes by targeting the root cause of the condition rather than just managing symptoms.
Spinraza belongs to a class of medicines known as antisense oligonucleotides, which work at the genetic level. SMA is caused by a mutation in the SMN1 gene, leading to insufficient production of the survival motor neuron (SMN) protein. Spinraza acts by modifying the splicing of the SMN2 gene, enabling the body to produce more functional SMN protein. This helps preserve motor neuron function, slow disease progression, and in many cases, improve motor milestones.
One of the key advantages of Spinraza is its proven clinical efficacy across all age groups, from infants to adults. Clinical studies have demonstrated improvements in muscle strength, motor function, and survival rates—especially when treatment is initiated early. Patients and caregivers often report enhanced quality of life, improved mobility, and increased independence over time.
Spinraza is administered via intrathecal injection (into the spinal canal) by a qualified healthcare professional. The treatment regimen begins with a loading phase (four doses within the first two months), followed by maintenance doses every four months. This structured dosing schedule ensures sustained therapeutic levels of the medication in the central nervous system.
From a safety perspective, Spinraza is generally well tolerated when administered under proper medical supervision. However, as with any advanced therapy, it requires regular monitoring, including platelet counts and kidney function tests, to ensure safe and effective use. Common side effects may include headache, back pain, and post-lumbar puncture discomfort. These are typically manageable and transient.
In terms of composition, Spinraza contains nusinersen sodium as its active ingredient, formulated in a sterile, preservative-free solution for injection. Each vial is carefully manufactured to meet strict pharmaceutical quality standards, ensuring consistency, purity, and reliability.
Storage and handling are critical for maintaining product integrity. Spinraza should be stored in a refrigerated environment (2°C to 8°C) and protected from light. It must not be frozen. When handled correctly, the product maintains its stability throughout its labeled shelf life, typically up to 36 months from the date of manufacture.
At Health Aid Pharmacy, we are committed to providing access to high-quality, authentic pharmaceutical products. Our sourcing standards ensure that every unit of Spinraza meets regulatory and safety requirements. For more information or to explore our full range of specialty medications, visit our homepage:
https://healthaidpharmacy.com/
Patients and healthcare providers worldwide trust Spinraza for its innovative mechanism, clinically proven outcomes, and long-term benefits. When sourced through a reliable provider, it represents a vital step forward in managing SMA and improving patient prognosis.






There are no reviews yet.